 Back in graduate school I used an experimental technique called Small Angle Neutron Scattering. Essentially, you take a beam of neutrons (say from a hole in a nuclear reactor, really!) and pass it through a test sample. A detector measures the scattering of neutrons by the samples. Analysis of the scattering pattern yields information about the microscopic structure of the sample.
Back in graduate school I used an experimental technique called Small Angle Neutron Scattering. Essentially, you take a beam of neutrons (say from a hole in a nuclear reactor, really!) and pass it through a test sample. A detector measures the scattering of neutrons by the samples. Analysis of the scattering pattern yields information about the microscopic structure of the sample.It turns out that different nuclei scatter neutrons differently, and you can use these differences to good effect in your experiments. An especially important difference is that between deuterium (a hydrogen atom with a nucleus that contains a proton and an extra neutron) and hydrogen. Since we studied aqueous dispersions of colloids and surfactants we would replace regular water (with hydrogen), with heavy water (with deuterium). Long nights (sometimes all night) watching the experiments gave us time to think.
We often wondered if drinking a bottle of heavy water would kill someone, or how much of the water in our bodies we could replace with heavy water without ill effects. Unlike other isotopic substitution, putting in deuterium for hydrogen actually has a chemical effect because you double the weight of the nucleus. Heavy water isn't radioactive so it wouldn't hurt you that way. We figured it would disrupt the action of important enzymes because the dielectric constant and solvent properties would be different.
An article in the Candian Journal of Physiology and Pharmacology referenced in Wikipedia actually explores some of these questions. Finally, answers for us, more than a decade later.
Bacteria and algae can handle 100% heavy water substituted for normal water.It turns out the symptoms of heavy water poisoning still looks like radiation damage, but that is because the enzyme effects we hypothesized above show up as eukaryotic cell's inability to repair DNA damage. It is too bad heavy water is expensive, $65 for 8 onces.
Protozoa can live in up to 70% heavy water.
Mammals 50% heavy water kills, 25% sterilizes, but 20% is survivable.
It would be an expensive way to poison someone, since you would need almost a quarter of the person's body weight to do it. I should have bet someone back in graduate school that I could drink a bottle and have it do no damage, because now I know it wouldn't. Too late.
 A recent article in Popular Science showing heavy water ice cubes (which are denser than normal water) sinking to the bottom of the glass reminded me of this discussion, and inspired the post.
A recent article in Popular Science showing heavy water ice cubes (which are denser than normal water) sinking to the bottom of the glass reminded me of this discussion, and inspired the post.tags: heavy water, neutron scattering, science, biology
7 comments:
I read that article too and wondered about it. Might have guessed you'd have a graph to illustrate it - always a pleasure to see your graphs.
If you've handled this stuff you can answer an obvious question I've often pondered. If you pick up a bottle of heavy water does it feel different? I mean is it like ordinary water but heavier? If so, is it an odd feeling?
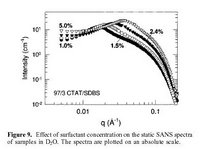
I must admit that the graph is from an old paper of mine in which I did some neutron scattering. I once was a scientist!
Heavy water is denser than water, but not that much denser than normal water, about 10% so you wouldn't really notice it.
More unexpected is holding bottles of two other clear liquids that look like water. Hexane is much lighter than water and you will feel it, and perhaps lift the bottle too far if you're expecting water. Fluorinated oils, say perfluorooctane, have flourine replacing the hydrogen hydrocarbon oils (octane). These are usually twice as dense as water and you get quite a shock when you pick one up and it is much heavier than expected.
Choosing the right chemical depends on the trick you want to pull.
I forget why I was researching this right now, but I found this blog on google and thought I'd add some "real world" experience (well, as real as my world goes, anyhow). I used to be a general construction contractor and had to know about this stuff when it regards plumbing.
You don't need a Norwegian dam to make heavy water, you can do it right at home! (please don't use this knowlege to make your own nukes, ok, you terrorist guys?)
It used to be common practice for plumbers to put a 'bumper' in water lines to eliminate 'water hammer', which is that jiddery shuddering thing faucets can do when you shut the tap off. It's easy for the plumber to fix by simply putting a 'T' in the line anywhere and running a stub of pipe from it that is capped and goes nowhere. Since the water can't fill the dead-end (when it is done right), it stays full of air and acts as a shock absorber for the whole building's system.
Problems occur (ie: dead people) when this dead-end stub fills with water and stays there. Don't ask me how, but this eventually becomes heavy water, and is toxic. Any building inspector will red-flag this before approving any plumbing job.
Also, the water in a recirculating hot water heating system (just like steam heat only with water), becomes heavy water over time. Once again; I have no idea why.
Please refer any technical questions to my friend and collegue, Professor Bongwater™, who can be reached at http://hogwhitman.com
Hog Whitman, I think what you are talking about it "Hard Water", which is just water that has very high mineral content.
The world health organization currently holds that there is insufficient evidence showing that hard water has any adverse effects on health. Though it does make soap difficult to lather!
well the steamer would make sense
since causing heavy water to vaporize would take more energy so the regular water would be vaporized first. Thus, overtime, deuterium collects.
http://articles.mercola.com/sites/articles/archive/2009/01/10/could-drinking-heavy-atoms-lengthen-your-life.aspx
This article says that drinking heavy water will increse your life span.
Post a Comment